The Complementary Glow
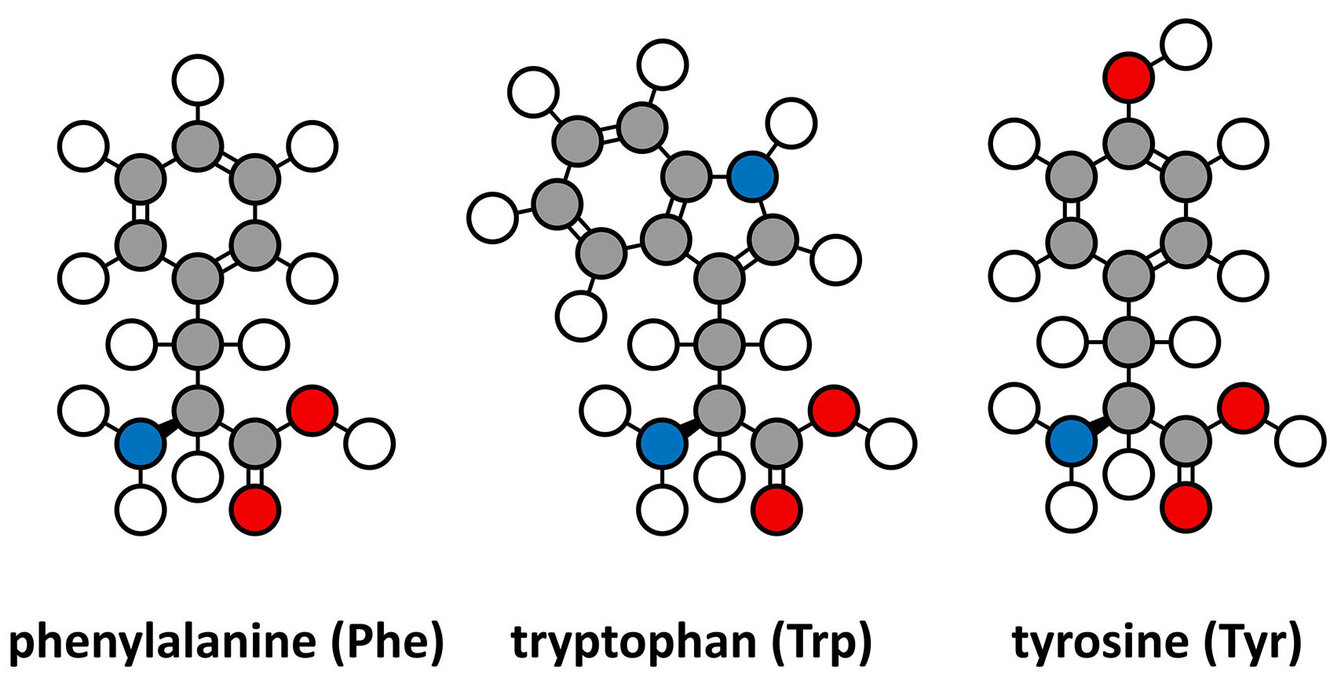
Proteins and their building blocks, amino acids, are too small to see with a standard microscope, so researchers use light to make them vibrate. By measuring these vibrations, they can identify exactly what a molecule is and how it is shaped. Traditional Raman spectroscopy uses a single photon of light to excite a molecule. The Optical Nanospectroscopy Group (Janina Kneipp) used a more advanced technique, so called SEHRS (Surface-Enhanced Hyper Raman Scattering) to find the spectra of tryptophan, phenylalanine and tyrosine. By using gold nanoparticles instead of the more common silver, the study paves the way for better biocompatibility in future medical and biological sensors. They specifically compared two types of citrate-stabilized gold nanoparticles (AuCit and AuAsc) and found that the way the nanoparticles were made significantly changed how the amino acids interacted with them. One of the most intriguing findings was the high variability in the spectra of tyrosine, which suggested that at certain concentrations, the molecules might be talking to each other through intermolecular interactions. Read the full article in Journal of Raman Spectroscopy for more information on SEHRS.
Abstract
Surface-enhanced hyper Raman scattering (SEHRS), the two-photon excited spontaneous Raman process that takes place in high local fields of plasmonic nanostructures, can provide information on molecular structure and interaction that is complementary to the information obtained by surface-enhanced Raman scattering (SERS). In this work, we report the nonresonant SEHRS spectra of the aromatic amino acids tryptophan, phenylalanine, and tyrosine using two types of citrate-stabilized gold nanoparticles. The SEHRS spectra are excited with a wavelength of 1064 nm and are compared to nonresonant SERS spectra, excited at a wavelength of 633 nm. Important bands in the spectra are assigned and reveal differences in the interaction of the amino acids with the gold nanostructures compared to previous reports of their spectra obtained with silver nanoparticles as plasmonic substrate. A greater variability of the SEHRS spectra of tyrosine with the similar gold substrates is found and discussed in the context of possible intermolecular interactions of the tyrosine molecule at the probed millimolar concentration. The nonresonant SEHRS spectra of the pure amino acids are a basis for probing the structure and interaction of whole proteins and supramolecular assemblies by this method using biocompatible gold nanoparticles rather than silver structures.